1. The Covid-19 Rapid Test is intended for professional use. It is not intended for self-testing or home use.
2. Any distributor should notify the local government of the product and obtain appropriate approval before importing the product and placing it on the local market.
3. we do not market, sell or distribute Covid-19 Rapid Test reagents in the United States that are not FDA approved.
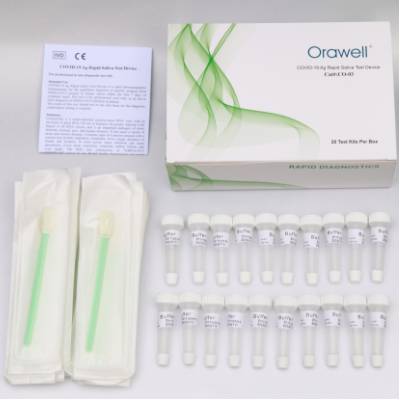
Covid-19 Rapid Test
On December 31, 2019, the first report of an ongoing outbreak of novel coronavirus disease was made in Wuhan, China. As of February 20, 2020, a cumulative total of 75,465 cases of COVID-19 have been reported in China.COVID-19 is transmitted by droplet and fecal transmission during close unprotected contact between an infected and infected person.
Since the beginning of the disease outbreak, our R&D team has been working on the development of a colloidal gold antibody test kit for COVID-19. Now, the Covid-19 rapid test kit can be used to qualitatively detect IgG/IgM antibodies to SARS-CoV-2 in human whole blood, serum or plasma. Our assay kit requires only 10ul of serum, plasma or whole blood, and results can be read in 10 minutes. It allows rapid screening of patients and reduces the false negative rate of PCR. This is very helpful in controlling the disease, reducing the spread of the virus and avoiding cross-infection. Since many areas are in flu season, we strongly recommend using our Covid-19 rapid test with the Accu-Tell® A&B flu test to quickly differentiate between the two diseases.
We are now ready to produce and offer this rapid test to our Covid-19 Rapid Test Distributors. Thanks to effective control measures, all of our employees and families are safe and healthy. Our daily office and daily production has been gradually resuming since mid-February.
If you need any information about our products or any information about the disease, please feel free to contact us.
Copyright © Jiangsu Well Biotech Co., Ltd. All Rights Reserved. |
Sitemap
| Powered by
苏ICP备18032586号