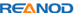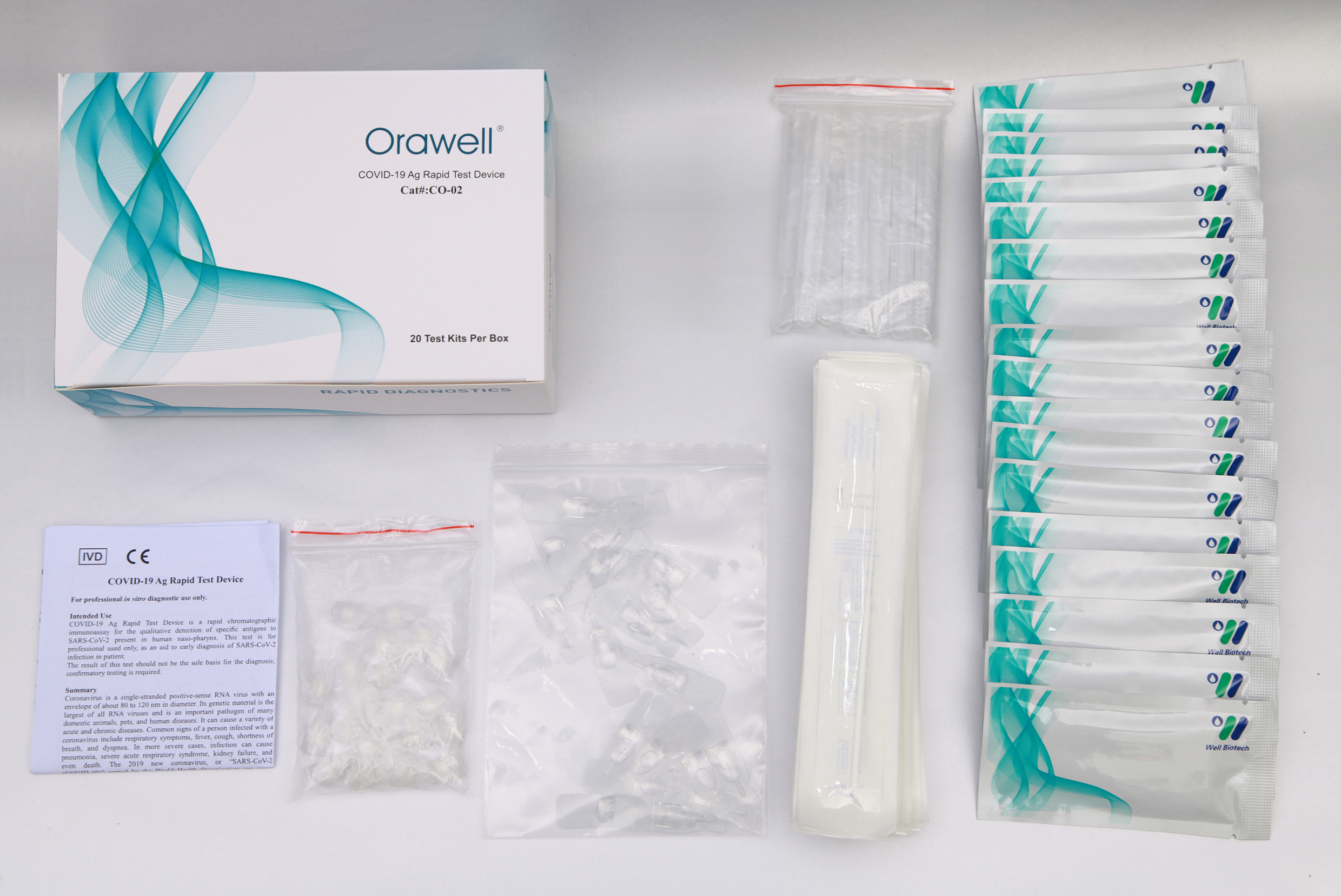
COVID-19 Ag Rapid Test Device is a rapid chromatographic immunoassay for the qualitative detection of N antigen to SARS-CoV-2 present in human nasal swabs. This test is for professional used only, as an aid to early diagnosis of SARS-CoV-2 infection in patient.
The result of this test should not be the sole basis for the diagnosis; confirmatory testing is required.
![]() Clinical Evalution Study for COVID-19 Ag.pdf
Clinical Evalution Study for COVID-19 Ag.pdf
![]() Instruction for Use-Ag NASAL.pdf
Instruction for Use-Ag NASAL.pdf
Product Details
The COVID-19 Ag Rapid Test Device uses double antibody sandwich immunoassay. The NC membrane pre-immobilized with monoclonal antibodies against SARS-CoV-2 antigen and anti-mouse polyclonal antibodies, and the colloidal-gold conjugated with monoclonal antibodies specific to SARS-CoV-2 antigen.
If SARS-CoV-2 antigen present in the sample, a complex formed between the anti-SARS-CoV-2 conjugate and the antigen will be caught by the specific anti- SARS-CoV-2 monoclonal coated on the T region. Results appear in 10 to 20 minutes in the form of a red line that develops on the strip.
Whether the sample contains the SARS-CoV-2 antigen or not, the solution continues to migrate to encounter another reagent (an anti-mouse IgG antibody) that binds the remaining conjugates, thereby producing a red line on the region C.
Kit Content
1). Test device (individually packed in a foil pouch.)
2). Extraction tube
3). Extraction buffer
4). Sterile swab
5). Instruction for use
Materials Required but not Provided
1. Timer 2. Transfer pipette
Performance Characteristics
Clinical Evaluation
Relative sensitivity: 126/133= 94.74% (95%CI 89.53%~97.43%)
Relative specificity: 200/200 >99% (95%CI 98.12%~100%)
Overall agreement: (126+200)/(126+0+7+200)*100%=97.90% (95%CI 95.73%~98.98%)
CI: Confidence Interval
 Dengue Rapid Tests (Dengue NS-1 Test/Dengue NS-1 Test)
Dengue Rapid Tests (Dengue NS-1 Test/Dengue NS-1 Test)
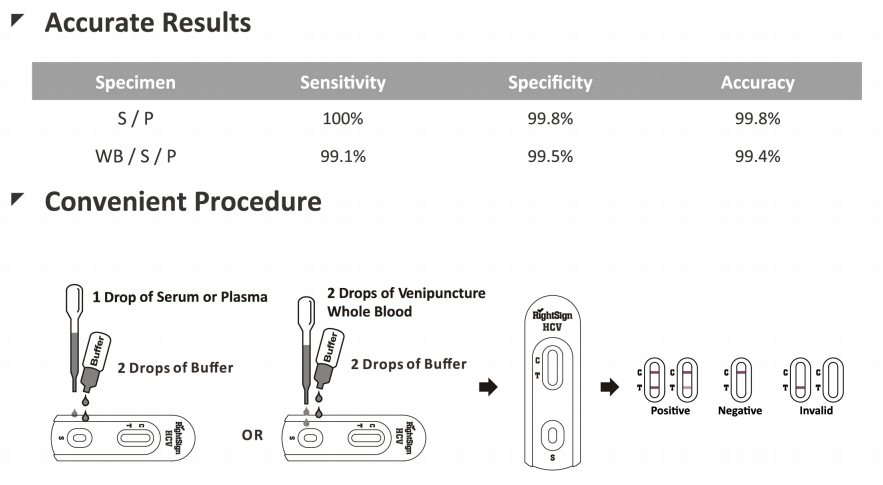 HCV Antibody Screen Test
HCV Antibody Screen Test
 H.pylori Rapid Tests(H. Pylori Ag Rapid Test/H. Pylori Ab Rapid Test)
H.pylori Rapid Tests(H. Pylori Ag Rapid Test/H. Pylori Ab Rapid Test)
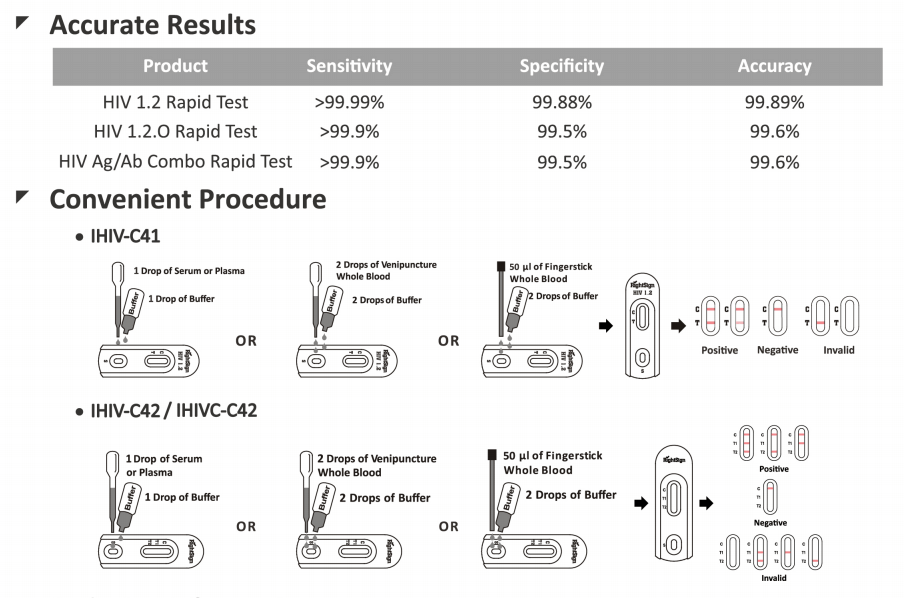 HIV ½ Antibodies Screen Test
HIV ½ Antibodies Screen Test
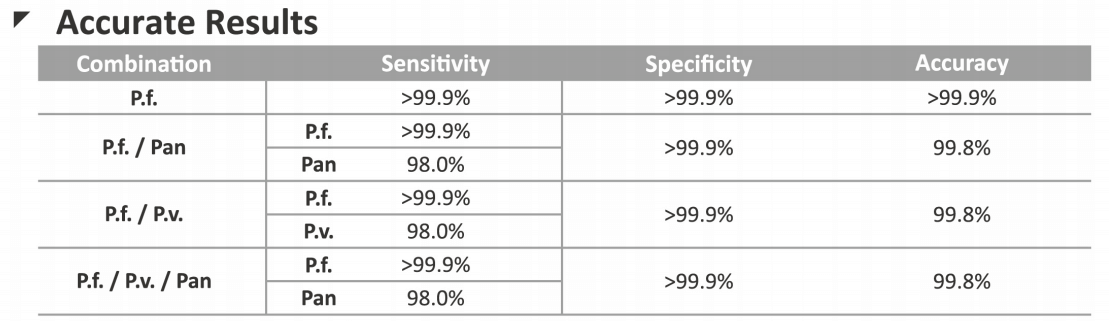 Malaria Rapid Tests (Malaria Pf/Pan Ag Test/Malaria Pf/Pv Ag Test)
Malaria Rapid Tests (Malaria Pf/Pan Ag Test/Malaria Pf/Pv Ag Test)
 Syphilis (TP antibody) Rapid Tests
Syphilis (TP antibody) Rapid Tests
 COVID-19 IgM/IgG Rapid Test (Colloidal Gold)
COVID-19 IgM/IgG Rapid Test (Colloidal Gold)
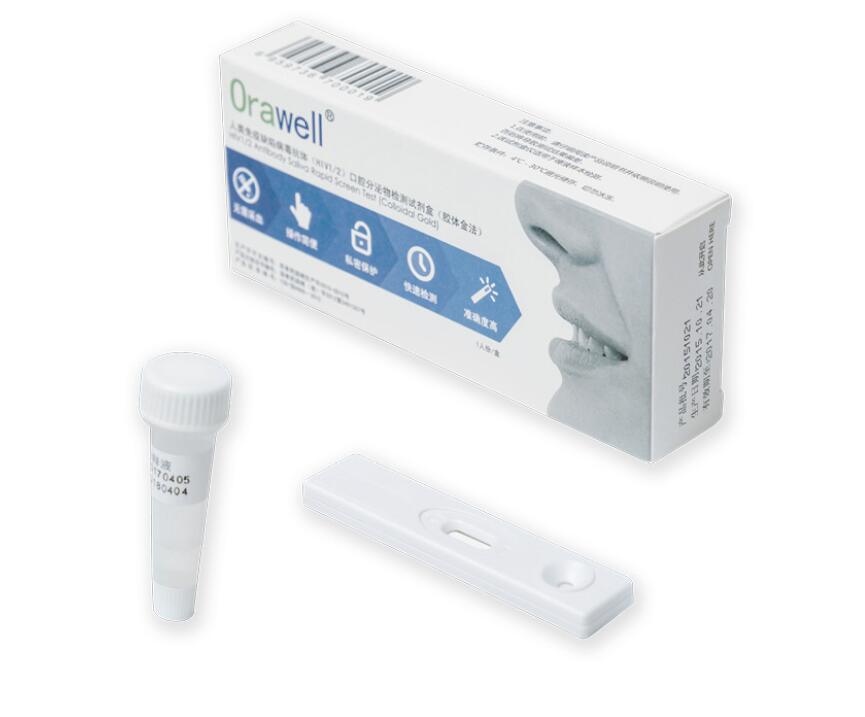 Oralwell® Oral Fluid HIV ½ Antibodies Screen Test
Oralwell® Oral Fluid HIV ½ Antibodies Screen Test
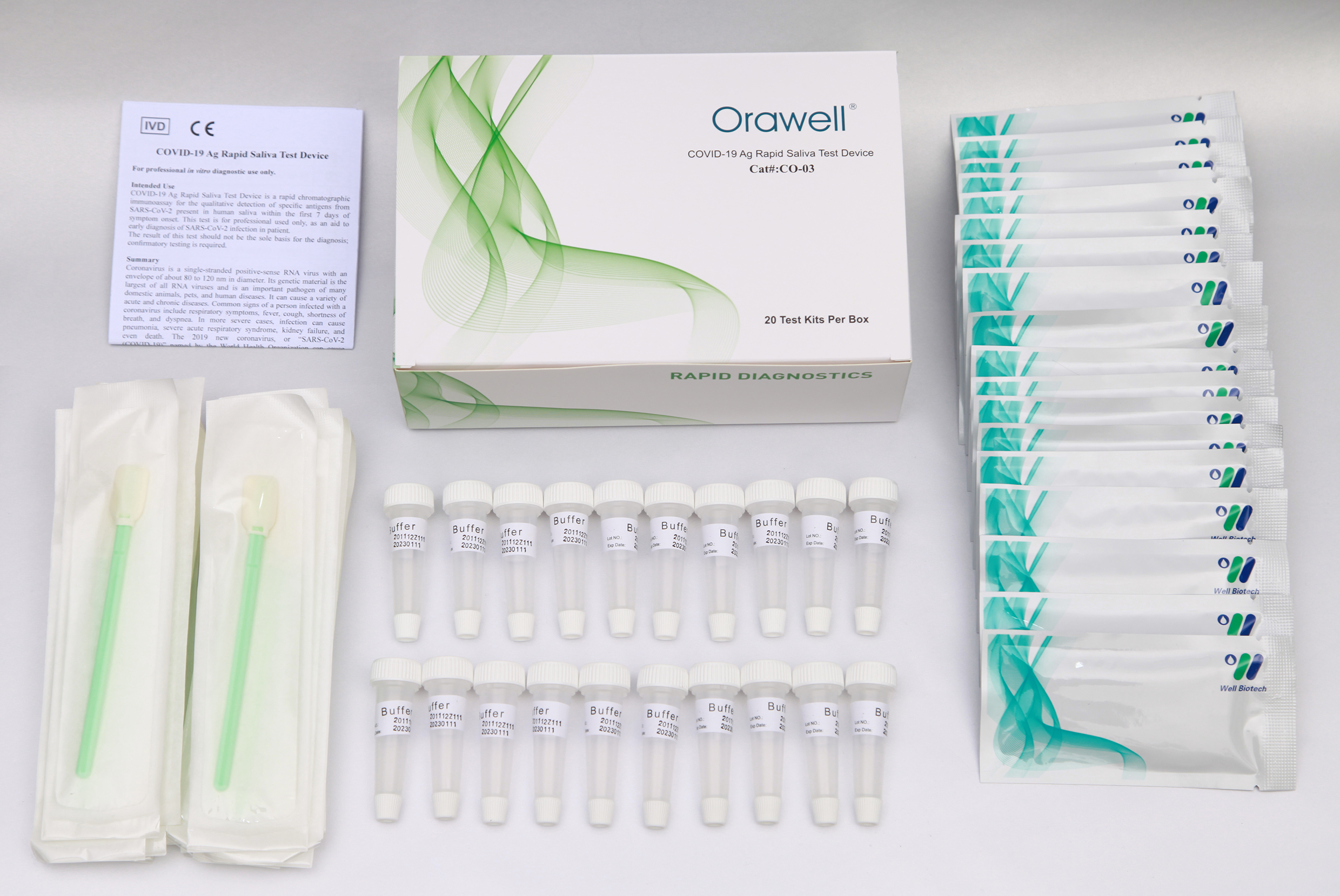 Orawell COVID-19 Ag Rapid Saliva Test Device
Orawell COVID-19 Ag Rapid Saliva Test Device
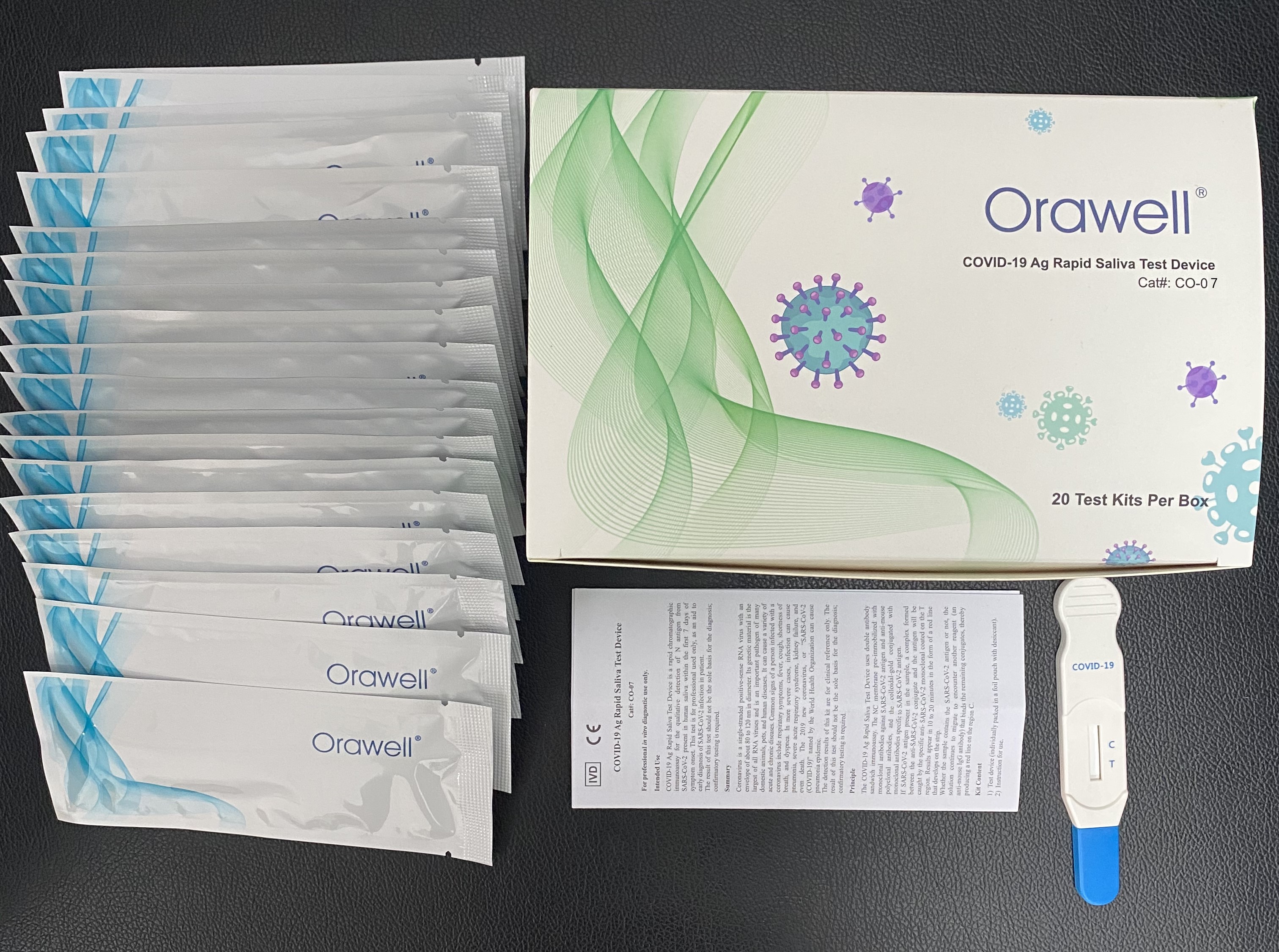 One Step COVID-19 Ag Rapid Saliva Test Device Lollipop
One Step COVID-19 Ag Rapid Saliva Test Device Lollipop
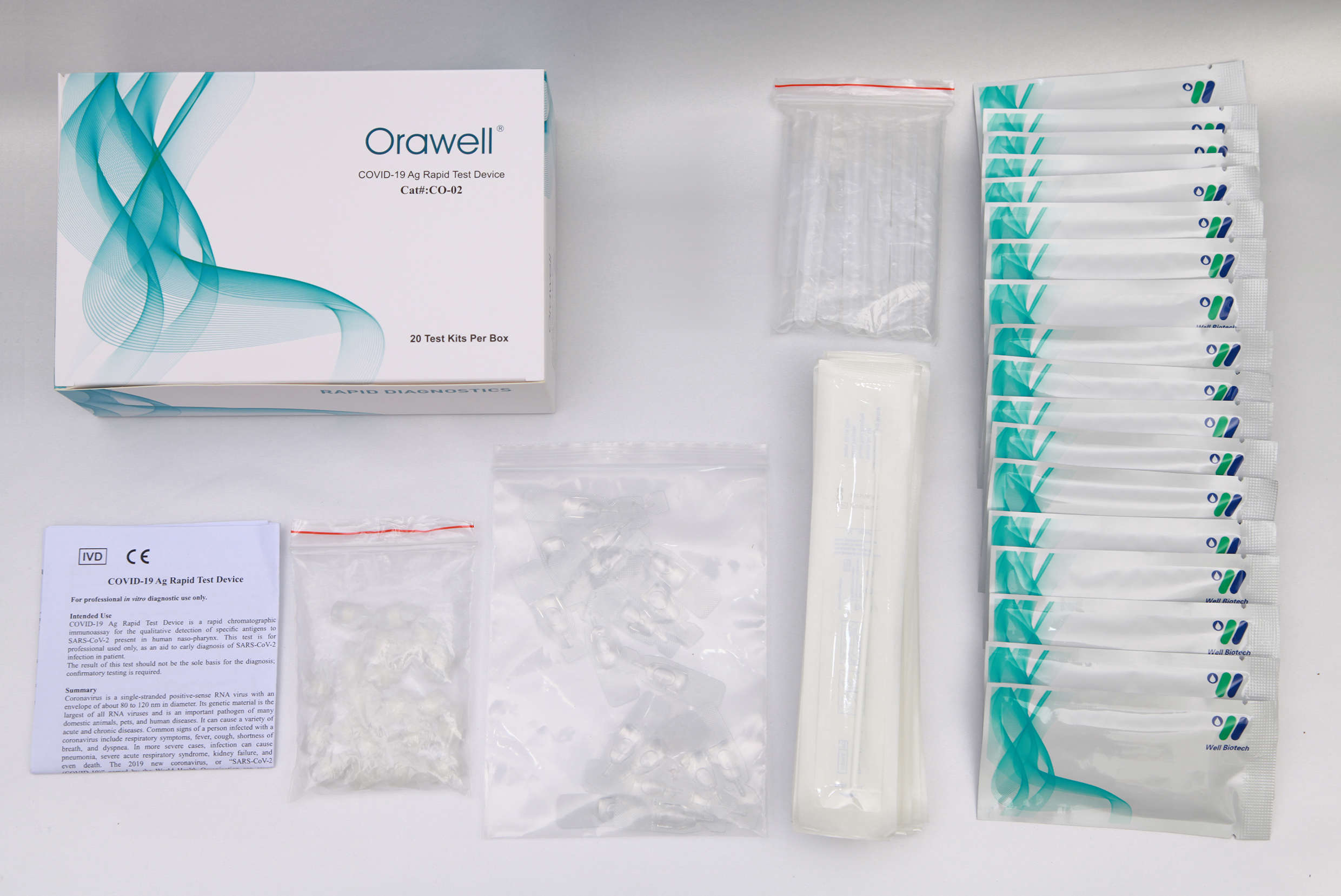 COVID-19 Ag Rapid Test Device
COVID-19 Ag Rapid Test Device
Copyright © Jiangsu Well Biotech Co., Ltd. All Rights Reserved. |
Sitemap
| Powered by
苏ICP备18032586号