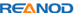The Zolpidem (ZOL) Rapid Test Cassette (Urine) is a rapid chromatographic immunoassay for the detection of Zolpidem Phenyl-4-carboxylic acid (ZOL metabolite) in urine at a cut-off concentration of 50 ng/mL. Gas chromatography/mass spectrometry (GC/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be applied to any drug of abuse test result, particularly when preliminary positive results are used.
Product Details
Zolpidem is a non-benzodiazepine hypnotic sold under the trade names Ambien®, Stilnox® and Edluar® for the treatment of insomnia. Zolpidem has not adequately demonstrated effectiveness in maintaining sleep, unless delivered in a controlled-release (CR) form. However, it is effective in initiating sleep. It works quickly, usually within 15 minutes, and has a short half-life of 2-3 hours. Because the characteristic of quick effect, low side effect, etc, Zolpidem has the trend of gradually replacing the barbiturates and benzodiazepine sleeping pills. The result of its widely used and easily obtained, the criminal cases showed a trend of rising. Zolpidem Phenyl-4-carboxylic acid is the major urinary metabolite of zolpidem, accounting for 51% of an administered dose. Literature references indicate the metabolite can be found in urine after ingesting a single therapeutic dose of zolpidem, for 2-3 days. Only 1% Zolpidem was extracted with original version by urine.
 Oxycodone (OXY) Rapid Tests
Oxycodone (OXY) Rapid Tests
 Phencyclidine (PCP) Rapid Tests
Phencyclidine (PCP) Rapid Tests
 Amphetamine (AMP) Rapid Tests
Amphetamine (AMP) Rapid Tests
 Methadone (MTD) Rapid Tests
Methadone (MTD) Rapid Tests
 Opioid (OPI) Rapid Tests
Opioid (OPI) Rapid Tests
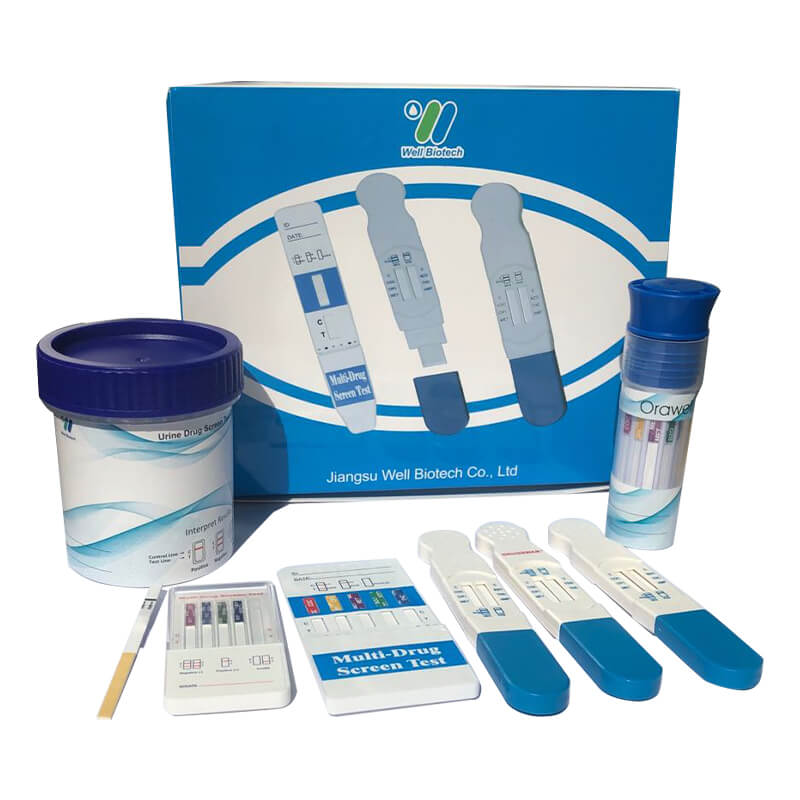 Alcohol Rapid Tests
Alcohol Rapid Tests
 Buprenorphine (BUP) Rapid Tests
Buprenorphine (BUP) Rapid Tests
 Benzodiazepines (BZO) Rapid Tests
Benzodiazepines (BZO) Rapid Tests
 Ethyl Glucuronide (ETG) Rapid Tests
Ethyl Glucuronide (ETG) Rapid Tests
 Zopiclone (ZOP) Rapid Tests
Zopiclone (ZOP) Rapid Tests
 Ketamine (KET) Rapid Tests
Ketamine (KET) Rapid Tests
 EMethadone Metabolite (EDDP) Rapid Tests
EMethadone Metabolite (EDDP) Rapid Tests
 Tricyclic Antidepressants (TCA) Rapid Tests
Tricyclic Antidepressants (TCA) Rapid Tests
 Methylphenidate (MPD) Rapid Tests
Methylphenidate (MPD) Rapid Tests
 Barbiturates (BAR) Rapid Tests
Barbiturates (BAR) Rapid Tests
 Propoxyphene (PPX) Rapid Tests
Propoxyphene (PPX) Rapid Tests
Copyright © Jiangsu Well Biotech Co., Ltd. All Rights Reserved. |
Sitemap
| Powered by
苏ICP备18032586号